|
Back to Blog
Nitrogen atomic radius5/17/2023 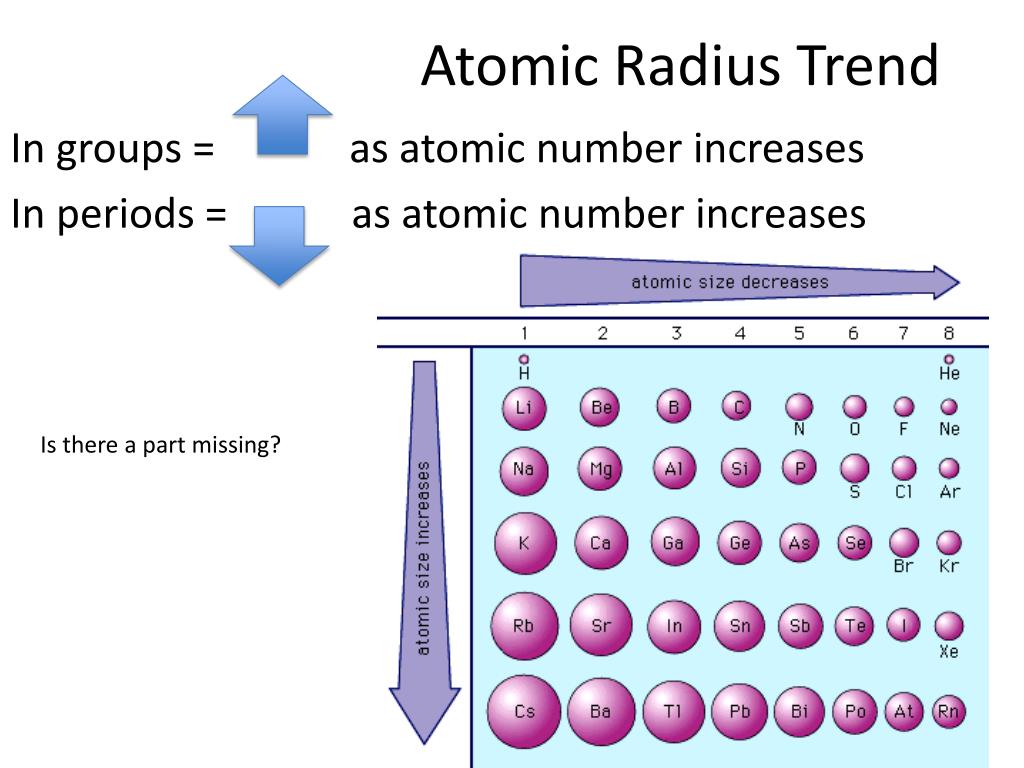
that for nitrogen, despite the trend in increasing IE1 values across a period. (Credit: Christopher Auyeung Source: CK-12 Foundation License: CC BY-NC 3. Within each period, the trend in atomic radius decreases as Z increases. Since it is sold as cylinders, a standard 50-liter one would cost you around $150 while the price of liquid nitrogen varies somewhere between $0.50 and $0.60 per gallon.\): Atomic radii of the representative elements measured in picometers. As you move down the nitrogen family: atomic radius increases, ionic radius increases, ionization energy decreases, and electronegativity decreases. Two of the electrons are in the s subshell, with 3 unpaired electrons in the p subshell. The 7 protons in a nitrogen atom are more effective at pulling in the two. Nitrogen is sometimes called ‘dephlogisticated’ or ‘burnt’ since once all the oxygen is taken out from the air it only comprises of the former. Nitrogen family elements consists of atoms having 5 electrons in their outer energy level. The trend for atomic radius in a vertical column is to go from smaller at the.During the Egyptian civilization, it used to be obtained by heating a mixture of dung, salt, and urine.It is a major constituent of proteins and nucleic acids (DNA and RNA) in the body of most living organisms.If the liquid form of the nonmetal comes in contact with the skin and eyes, it may cause some dangerous effects like frostbite, pain, and burns. Deep-sea divers are prone to a condition called narcosis marked by symptoms like hallucination, disorientation, loss of concentration, and short-term memory loss as a result of increased nitrogen pressure in the blood. Inhalation of high levels of N 2 may lead to suffocation, weakness, and unconsciousness. Nitrogen mustard, a type of mustard gas comprising of nitrogen, are used as chemotherapeutic agents in the treatment of cancer. Not only does the size (radius) of the atoms decrease (from 70 pm for C to 38 pm for Ne), so does the length of the bonds between the atoms and hydrogen (from.The number of shells increases hence screening effect increases. Explanation: As we move down the group atomic number increases, hence the nuclear charges increases. Nitrogen plastic welders are used in repairing plastic components of automobiles by eliminating oxygen. Periodic Trend in Atomic Radius Along the Group: Trend: The atomic size increases down the group. 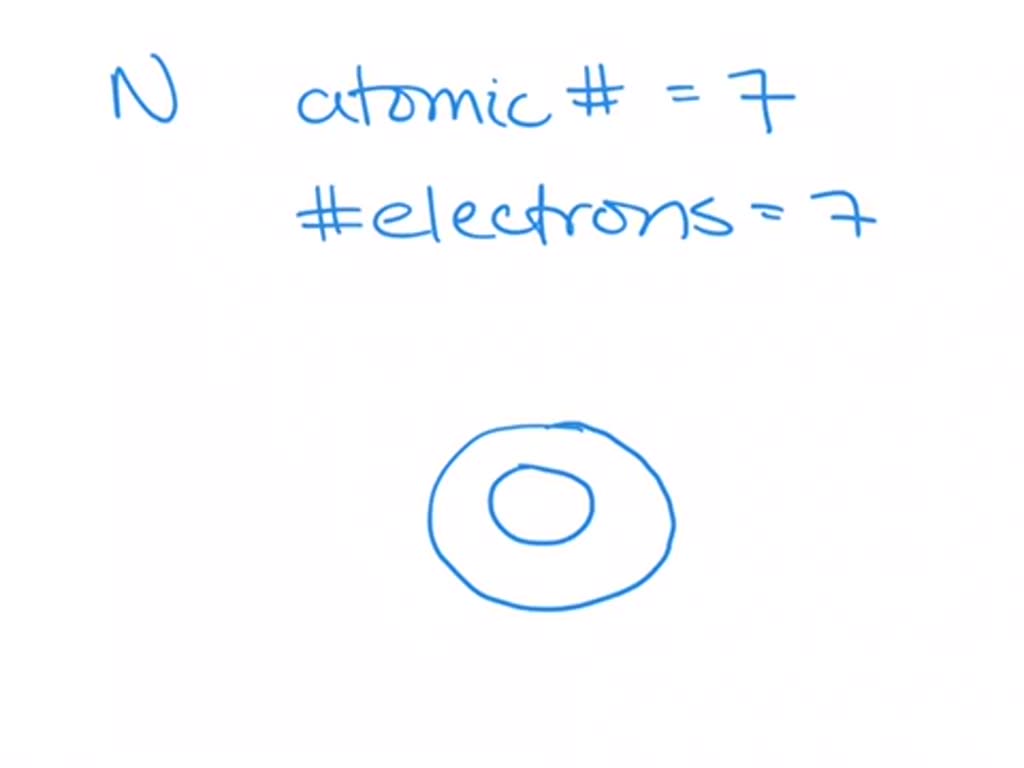
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed

